
He concludes that atoms of different weights combine differently, according to specific laws of proportion, to form the different elements. His chemical atomic theory seeks, among other things, to establish how the atoms of different elements combine to form compounds. Dalton claims that, since elements are composed of atoms and since there are differences between elements, there must also be differences between the atoms that compose those elements. For Dalton, the primary determinable feature of chemical atoms is their weight. 
Dalton purports to establish that chemical atoms are empirical entities, that is, that they have empirical and quantifiable features that can be experimentally determined. The challenge for Dalton, however, is to avoid any metaphysical implications in his atomism by employing the experimental and quantitative criteria advanced by Lavoisier. John Dalton, on the other hand, seeks to establish an empirical link between ‘elements’ and ‘atoms’, through the notion of ‘chemical atom’. 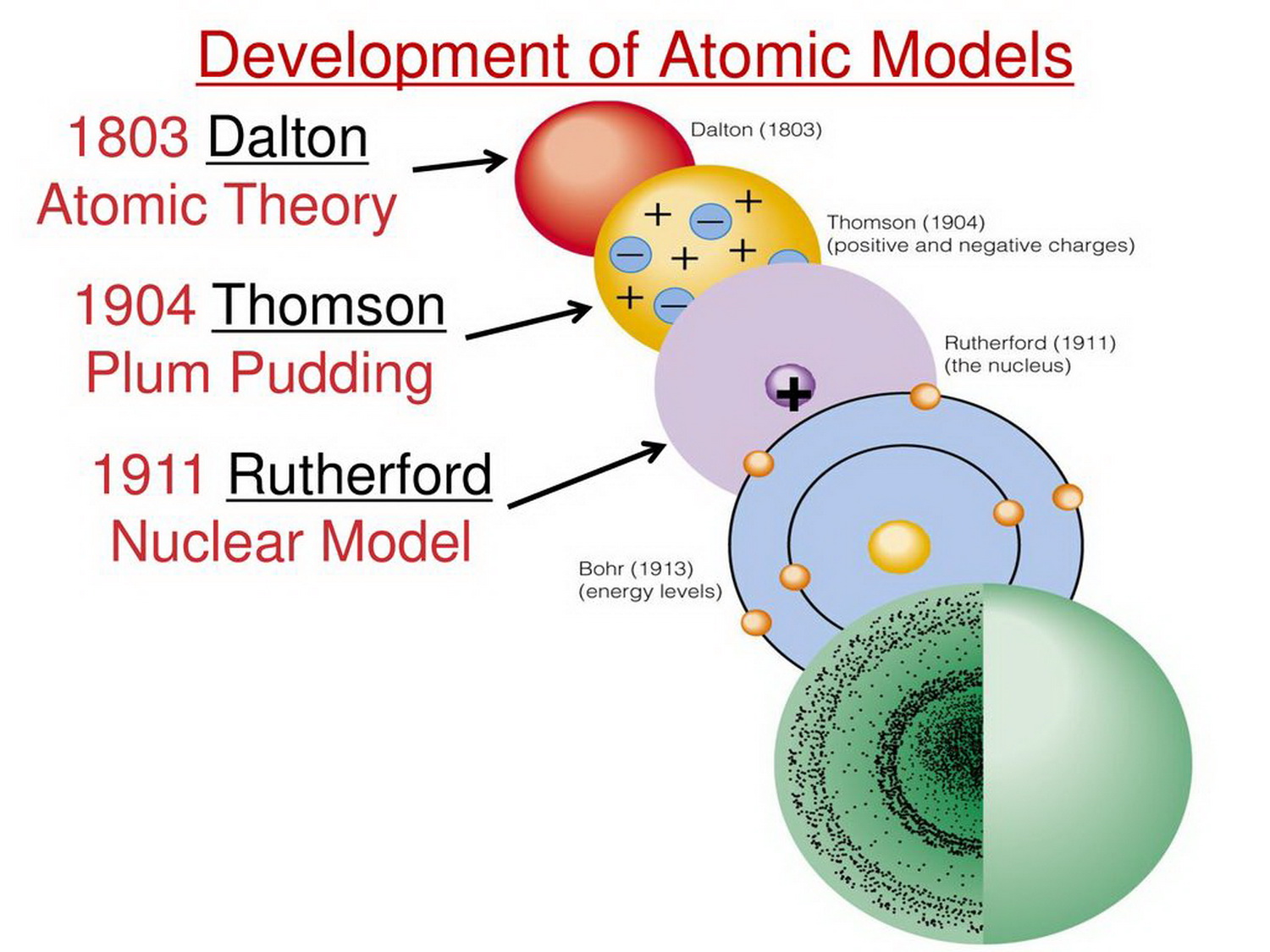
Instead, by ‘element’, Lavoisier means those substances that remain as the last product of chemical analysis. Thus, for Lavoisier, the term ‘element’ should not be applied to atoms or to fundamental particles, which he considers to be suspect metaphysical entities. In his view, since atoms have no empirically determinable or quantifiable properties, they contribute nothing to actual experimental work or to the chemist’s understanding of chemical elements. His emphasis on both empirical data and quantitative analysis greatly influences his position on this issue. Lavoisier rejects the epistemic value of such positing and considers it to be mere metaphysical speculation. Modern scientists have found smaller particles that make up the protons, neutrons, and electrons, although the atom remains the smallest unit of matter that can't be divided using chemical means.This paper addresses the fundamental disagreement between the views of Antoine Lavoisier and John Dalton regarding the scientific and epistemic value of positing indivisible atoms as the most simple and fundamental particles of matter. For atoms with a high number of electrons, relativistic effects come into play, since the particles are moving at a fraction of the speed of light. Rather than the circular orbits of Rutherford's model, modern atomic theory describes orbitals that may be spherical, dumbbell-shaped, etc. :max_bytes(150000):strip_icc()/atomic-structure-conceptual-artwork-99312661-58af58c75f9b5860467ff472.jpg)
The electron can potentially be found anywhere in the atom but is found with the greatest probability in an atomic orbital or energy level. Quantum mechanics led to an atomic theory in which atoms consist of smaller particles. This, in turn, led to Werner Heisenberg's uncertainty principle (1927), which states that it's not possible to simultaneously know both the position and momentum of an electron. Louis de Broglie proposed a wavelike behavior of moving particles, which Erwin Schrödinger described using Schrödinger's equation (1926). In 1913, Frederick Soddy described isotopes, which were forms of an atom of one element that contained different numbers of neutrons. Several discoveries expanded the understanding of atoms. The model and its validation in 1908 by Jean Perrin supported atomic theory and particle theory.īohr's model explained the spectral lines of hydrogen but didn't extend to the behavior of atoms with multiple electrons. In 1905, Albert Einstein postulated that Brownian motion was due to the movement of water molecules. Avogadro's law made it possible to accurately estimate the atomic masses of elements and made a clear distinction between atoms and molecules.Īnother significant contribution to atomic theory was made in 1827 by botanist Robert Brown, who noticed that dust particles floating in water seemed to move randomly for no known reason. In 1811, Amedeo Avogadro corrected a problem with Dalton's theory when he proposed that equal volumes of gases at equal temperature and pressure contain the same number of particles. His oral presentation (1803) and publication (1805) marked the beginning of the scientific atomic theory. He proposed that each chemical element consists of a single type of atom that could not be destroyed by any chemical means. Dalton's law of multiple proportions drew from experimental data. These theories didn't reference atoms, yet John Dalton built upon them to develop the law of multiple proportions, which states that the ratios of masses of elements in a compound are small whole numbers. Ten years later, Joseph Louis Proust proposed the law of definite proportions, which states that the masses of elements in a compound always occur in the same proportion. In 1789, Antoine Lavoisier formulated the law of conservation of mass, which states that the mass of the products of a reaction is the same as the mass of the reactants. It took until the end of the 18th century for science to provide concrete evidence of the existence of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
